1. Introduction
Stenotrophomonas maltophilia (formerly Pseudomonas maltophilia and Xanthomonas maltophilia) is a Gram-negative, non-fermentative bacillus that causes opportunistic infections with a wide range of clinical presentations, most commonly presenting as pneumonia and bacteremia (Abbott et al., 2011; Brooke, 2012). It is reported as the third most common non-fermenting bacillus causing nosocomial infections, after Pseudomonas aeruginosa and Acinetobacter baumannii, although rare cases of community-acquired infections have also been noted (Brooke, 2012; Falagas et al., 2009; Trends & Associated, 2019). During the COVID-19 pandemic, this infectious agent was identified as one of the most common causes of respiratory superinfections in patients with a severe clinical course (Mikhailovich et al., 2024). Risk factors associated with S. maltophilia infection include cystic fibrosis, various immunodeficiency states, immunosuppressive therapy, and prolonged hospitalization (Çıkman et al., 2016; Hafiz et al., 2022; Mojica et al., 2022).
S. maltophilia is a multidrug-resistant (MDR) organism (Brooke, 2012). Resistance mechanisms, including the production of beta-lactamases, efflux pumps, and modification of antibiotic target sites, significantly narrows the therapeutic spectrum for infections caused by this bacterium (Brooke, 2012; Trends & Associated, 2019). The first-line treatment for S. maltophilia infections is trimethoprim–sulfamethoxazole (Juhász et al., 2015). In cases of resistance to this antibiotic, alternative agents such as levofloxacin, ceftazidime, tetracyclines, and colistin can be used (Çıkman et al., 2016; Mojica et al., 2022). Recent guidelines from the Infectious Diseases Society of America (IDSA) recommended two combination therapy protocols: 1) combination of two following agents: cefiderocol, minocycline, trimethoprim-sulfamethoxazole, or levofloxacin; and 2) combination of ceftazidime-avibactam with aztreonam (Fan et al., 2025) . However, strains resistant to all drugs of choice have been reported (Çıkman et al., 2016; Juhász et al., 2015; Mojica et al., 2022).
Although S. maltophilia is not considered a highly virulent pathogen, infections caused by this organism are associated with considerable morbidity and mortality (Brooke, 2012; Cillóniz et al., 2019; Gibb & Wong, 2021). The ability to form biofilms plays a crucial role in the pathogenesis of S. maltophilia infections, particularly in hospital setting. The organism readily adheres to both biological and abiotic surfaces, which explains its frequent association with infections related to medical devices and mechanical ventilation (Brooke, 2012; Hafiz et al., 2022). Biofilm formation is closely linked to increased tolerance to environmental stressors, reduced susceptibility to phagocytic activity and other host immune defense mechanisms, as well as decreased susceptibility to antimicrobial agents (Abbott et al., 2011; Mikhailovich et al., 2024). Furthermore, biofilms may facilitate the development of antimicrobial resistance through horizontal gene transfer among bacterial cells embedded within the biofilm matrix (Flores-Treviño et al., 2019).
The objectives of this study were to evaluate the antimicrobial susceptibility of clinical S. maltophilia isolates to trimethoprim–sulfamethoxazole, ceftazidime, levofloxacin, chloramphenicol, and colistin, and to assess their biofilm production capacity.
2. Material and methods
2.1. Bacterial collection
Clinical isolates of S. maltophilia from the collection of the Institute of Microbiology and Immunology of the Faculty of Medicine, University of Belgrade, collected from 2017 to 2022, were included in this study. After arriving at the bacteriological laboratory, the isolates were cultured on blood agar (Promedia, Serbia) and preserved in skim milk broth (HiMedia, India) at -80°C. Identification was confirmed using MALDI-TOF MS (Matrix assisted laser desorption/ionization-time of flight mass spectrometry, Bruker, Germany).
2.2. Antimicrobial susceptibility of S. maltophilia isolates
Disc diffusion test was used to assess susceptibility to trimethoprim-sulfamethoxazole (1.25μg+23.75μg) and levofloxacin (5μg) (Bio-Rad, USA). The susceptibility to ceftazidime and chloramphenicol was examined by gradient test (Liofilchem, Italy) . The broth microdilution method (Liofilchem, Italy) was used to test the colistin sensitivity according to the manufacturer’s instructions. The results were interpreted according to the European Committee on Antimicrobial Susceptibility Testing (EUCAST) standard for trimethoprim-sulfamethoxazole and to the Clinical and Laboratory Standards Institute (CLSI) standard for levofloxacin, chloramphenicol, and ceftazidime (CLSI M100-ED35:2025, 2025; EUCAST, 2025). For colistin, CLSI breakpoints for Pseudomonas aeruginosa were used with susceptibility cutoffs of 2 μg/mL (CLSI M100-ED35:2025, 2025; Yero et al., 2020).
2.3. Biofilm producing capacity
Biofilm producing capacity was tested using the previously described method by Stepanović et al (Stepanović et al., 2007). In a microtiter plate with 96 wells, a bacterial suspension with a concentration of 106 CFU/ml Mueller-Hinton broth (Oxoid, UK) was inoculated. Biofilm production assays were performed in triplicate. After 24 hours at 37°C, the wells were washed three times with sterile physiological solution. After biofilm fixation by methanol, staining was performed using 1% crystal violet. The dye bound to the bacterial cells was redissolved by 96% ethanol and optical density (OD) was measured at 570 nm using a microplate reader (Multiska FC Microplate Photometer, Thermo Scientific, USA). The categories of biofilm formation were interpreted according to the standards established by Stepanović et al and the strains were classified into four groups: no-biofilm producers, weak biofilm producers, moderate biofilm producers and strong biofilm producers (Stepanović et al., 2007).
2.4. Statistical analysis
Statistical data analysis was performed using EZR software (R Commander Version 2.7-1). Fisher’s test was used to assess the existence of a statistically significant relationship between the ability to produce biofilm and the existence of resistance to certain antibiotics or the origin of certain isolates.
3. Results and discussion
We included 24 isolates of S. maltophilia. The strains were isolated from patients with an average age of 65.2±17.6 years. Males (N=13, 54.2%) were slightly more prevalent than females (N=11, 45.8%). Most isolates (N=7/24, 29.1%) came from medical implants (central venous catheter-CVC, chest drain, aspiration catheter). An equal number of strains was isolated from samples from the respiratory tract (tracheal swab, bronchoalveolar lavage-BAL, tracheal aspirate; N=6/24, 25%) and blood (N=6/24, 25%). Other strains were isolated from wound swabs (N=3/24, 12.5%) and urine (N=2/24, 8.3%). Using MALDI-TOF MS, the identification of all isolates included in the research was confirmed.
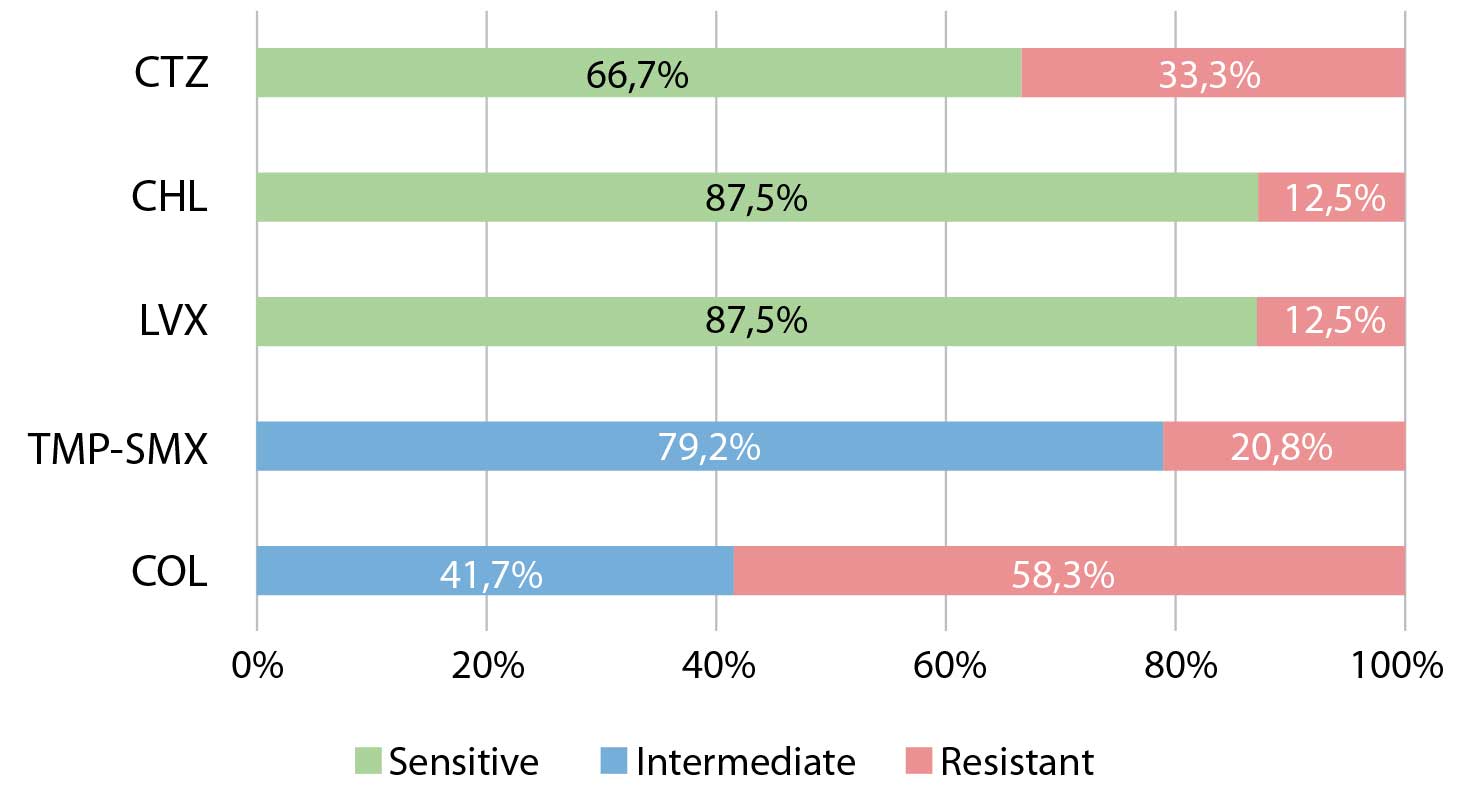
Figure 1. Susceptibility of clinical isolates of Stenotrophomonas maltophilia to selected antibiotics
TMP-SMX – trimethoprim-sulfamethoxazole; LVX – levofloxacin; CTZ – ceftazidime; CHL: chloramphenicol; COL: colistin
The results of the antibiogram are presented in Figure 1. The research detected three S. maltophilia isolates (12.5%) resistant to all tested antibiotics except colistin. Two out of three isolates (66.7%) with colistin as only therapeutic option were isolated from medical implants. The reduced sensitivity of S.maltophilia isolates to broad-spectrum of antibiotics is mostly mediated by chromosomally encoded mechanisms, which confirms the phenotypic similarity between clinical and environmental isolates (Gil-Gil et al., 2020; Wang et al., 2018)in close association with soil, sewage, and plants. Stenotrophomonas maltophilia , the first member of this genus, is the predominant species, observed in soil, water, plants, animals, and humans. It is also an opportunistic pathogen associated with the increased number of infections in both humans and animals in recent years. In this article, we summarize all Stenotrophomonas species (mainly S. maltophilia. The similarity of the MDR region of S.maltophilia with the plasmid and chromosomal sequences of other Gram-negative bacteria, such as Pseudomonas spp, Salmonella spp, and Escherichia coli, were observed, which indicates the acquisition of resistance mechanisms through horizontal gene transfer (Wang et al., 2018). An increase in the frequency of multiresistant strains (11-31%) was recorded, in which resistance to trimethoprim-sulfamethoxazole also appears more frequently (Mojica et al., 2022). Due to high incidence of inherited antibiotics resistance, the breakpoints for S. maltophilia isolates are available for a limited number of antibiotics (CLSI M100-ED35:2025, 2025; EUCAST, 2025).
Trimethoprim-sulfamethoxazole is considered the most effective antibiotic in the treatment of S. maltophilia infections, although an increasing number of studies noted the presence of resistance to this antibiotic (Chang et al., 2015; Gil-Gil et al., 2020; Wang et al., 2018)in close association with soil, sewage, and plants. Stenotrophomonas maltophilia , the first member of this genus, is the predominant species, observed in soil, water, plants, animals, and humans. It is also an opportunistic pathogen associated with the increased number of infections in both humans and animals in recent years. In this article, we summarize all Stenotrophomonas species (mainly S. maltophilia. A study from Saudi Arabia registered a lower frequency of resistance to trimethoprim-sulfamethoxazole (4.1%) compared to our result (20.8%) (Hafiz et al., 2022). Studies conducted in the United Kingdom (12.7%) and Hungary (2.5%) also recorded a lower frequency of resistance to this antibiotic in S. maltophilia (Juhász et al., 2015; Milne & Gould, 2012). In the period 2018-2020 in Mexico, resistance to trimethoprim-sulfamethoxazole of 44% among planktonic and 100% among biofilm isolates was recorded (José Mauricio Del Río-Chacón et al, 2024). The study carried out in 2016 in Belgrade in pediatric patients didn’t registered isolates resistant to this antibiotic (Madi et al., 2016)which has recently been recognized as a globally multi-drug resistant organism. The aim of this study was genotyping and physiological characterization of Stenotrophomonas maltophilia isolated in a large, tertiary care pediatric hospital in Belgrade, Serbia, hosting the national reference cystic fibrosis (CF. The carrier of resistance to trimethoprim-sulfamethoxazole is considered to be the sul1 gene located on class 1 integrons, as well as the sul2 gene located on the plasmid or in the chromosomal DNA of the bacteria (Toleman et al., 2007). In recent years, the development of resistance has been linked to SmeDEF efflux pumps and the dfrA gene, which encodes the enzyme dihydrofolate reductase (Chang et al., 2015; Sánchez & Martínez, 2015).
The increase in trimethoprim-sulfamethoxazole resistance rates indicates that empirical therapy with this drug is not sufficient for complete cure, so alternative therapeutic options are considered.(Chung et al., 2015)sul2, and sul3 genes, integrons, insertion sequence common region (ISCR. Levofloxacin is one of the alternative choices in the treatment of S. maltophilia infections (Juhász et al., 2014). Our study observed a slightly lower frequency of resistance to this antibiotic (12.5%) compared to the results recorded in Hungary (20%) and the United Kingdom (37.5%) (Juhász et al., 2015; Milne & Gould, 2012). Besides the mutations in genes encoding topoisomerases (gyrB, parC, parE), the main role in the development of resistance to levofloxacin and other fluoroquinolone is mediated by the Smqnr gene, which protect topoisomerases from quinolones, and hyperproduction of efflux pumps (SmeDEF, SmeVWX), which are associated with intrinsic or acquired resistance (García-León et al., 2015; Sánchez & Martínez, 2010; Wang et al., 2018). Levofloxacin has been shown to inhibit biofilm formation and maturation, which increases its clinical value in the treatment of S. maltophilia infections, especially infections of the respiratory tract (Flores-Treviño et al., 2019; Juhász et al., 2014).
This study identified 33.3% of isolates resistant to the ceftazidime. By MIC testing, MIC50 (6 µg/ml) and MIC90 (≥256 µg/ml) values were determined. Resistance to chloramphenicol was observed in 12.5% (N=3/24) of cases. It has been shown that the MIC50 for chloramphenicol is 3 µg/ml, and the MIC90 is ≥256 µg/ml.
Despite the innate resistance to beta-lactam antibiotics, ceftazidime shows in vitro activity against S. maltophilia, based on inhibitory action to L2-producing strains, therefore it is the drug of choice due to the inability to use trimethoprim-sulfamethoxazole (Abbott et al., 2011; Lin et al., 2021). Our study recorded significantly lower results compared to the study conducted in Turkey (72%) (Çıkman et al., 2016). The increase of resistance rate to ceftazidime is proved by the results of research from Saudi Arabia and Greece, where the resistance rate was 62.1% and 73.5%, respectively (Hafiz et al., 2022; Samonis et al., 2012)that infects critically ill patients and has expressed resistance against antimicrobial therapy. The aim of this study was to examine the epidemiological pattern, resistance characteristics and clinical outcomes of S. maltophilia infections in hospitalized patients. Methods. The study included 393 S. maltophilia isolates from different clinical specimens as well as the clinical data of 209 Intensive Care Unit (ICU. All isolates included in Hungarian study from 2015 were resistant to this drug, and values of MIC50 (128mg/l) and MIC90 (512mg/l) were higher compared to our results (Juhász et al., 2015). Due to the resistance associated with L1 and L2 beta-lactamases, the beneficial therapeutic effect of ceftazidime on S.maltophilia is achieved by combined therapy (Abbott et al., 2011). The combination of ceftazidime-avibactam and aztreonam showed great in vitro efficiency (Gibb & Wong, 2021). Numerous studies point out that aztreonam-avibactam has the strongest in vitro therapeutic effect on S.maltophilia strains compared to other related agents, and since 2024 this agent has been approved by the European Medicines Agency for the treatment of infections caused by aerobic Gram-negative bacilli in people with limited therapeutic options (Lin et al., 2021; Sader et al., 2020, 2025).
This study obtained lower resistance rate of chloramphenicol (12.5%) compared to research conducted in Turkey (18.2%) (Çıkman et al., 2016). Juhász et al. (Juhász et al., 2015) recorded a higher frequency of resistance (44%) in Hungary. The sensitivity to this antibiotic was 83.8% in Greece (Samonis et al., 2012)Crete, Greece, between 1/2005-12/2010. S. maltophilia antimicrobial susceptibility was tested with the agar dilution method. Prognostic factors for all-cause in-hospital mortality were assessed with multivariate logistic regression. Results: Sixty-eight patients (median age: 70.5 years; 64.7% males. A study conducted in the USA in 2020, in which 51.2% of resistant isolates were confirmed, reported higher values of MIC50 (16mg/l) and MIC90 (≥64mg/l) compared to our results (Biagi et al., 2020). The low rate of resistance of S. maltophilia to chloramphenicol is associated with its limited use due to its myelotoxic effect (Abbott et al., 2011). The main carrier of chloramphenicol resistance in S. maltophilia strains is floR gene, which encodes an exporting protein, as well as acetyltransferase genes catB2 and catB8 (Wang et al., 2018).
There are no standards and recommendations for the determination of sensitivity to colistin in S. maltophilia isolates. However, the in vitro activity of colistin against S. maltophilia has been observed and it can be used in combination with other antibiotics as a drug of last resort in the therapy of these infections (Gülmez et al., 2010)E-test, Phoenix system, and reference agar dilution method and also to evaluate the in vitro activity of various antimicrobial combinations against multidrug-resistant S. maltophilia. Susceptibilities to several antimicrobial agents were determined by agar dilution, disc diffusion, and E-test according to the US Clinical Laboratory and Standards Institute (CLSI. Slightly more than half of the isolates included in our research (N=14/24, 58.3%) showed resistance to colistin, and this high frequency of resistance is in agreement with the results of numerous studies (Galani et al., 2008; Moskowitz et al., 2010; Rodríguez et al., 2014)whilst it was less active both against Enterobacter spp. and Klebsiella pneumoniae (MIC for 50% of the organisms (MIC50. MIC50 (8 µg/ml) and MIC90 (≥16 µg/ml) for colistin were determined. Research conducted in Spain in period 2014-2018. recorded lower incidence of resistant strains 28.3% and values of MIC50 (2 µg/ml) and MIC90 (8 µg/ml) compared to our results (Cercenado et al., 2021).
Due to innate resistance to a large number of antibiotics, as well as the reduction of sensitivity to drugs already used to treat infections, S. maltophilia, together with other Gram-negative non-fermentative bacilli, is high-priority pathogen for the development of new therapeutic options (Závora et al., 2025). Cefiderocol is a novel parenteral antibiotic from the group of cephalosporins which is approved for the treatment of infections in adults with limited therapeutic options in Europe and the USA due to its favorable in vitro effect on Gram-negative non-fermenting bacilli, (Cercenado et al., 2021; Katsube et al., 2019; McCreary et al., 2021)pharmacokinetic/pharmacodynamic breakpoints were used. Results: Of 2303 isolates [1502 (65.2%. Despite the fact that one resistant strain was detected in Japan by microbroth dilution test, cefiderocol has the potential to become an empiric therapy for infections caused by S. maltophilia strains due to its high efficacy (Aoki et al., 2025).
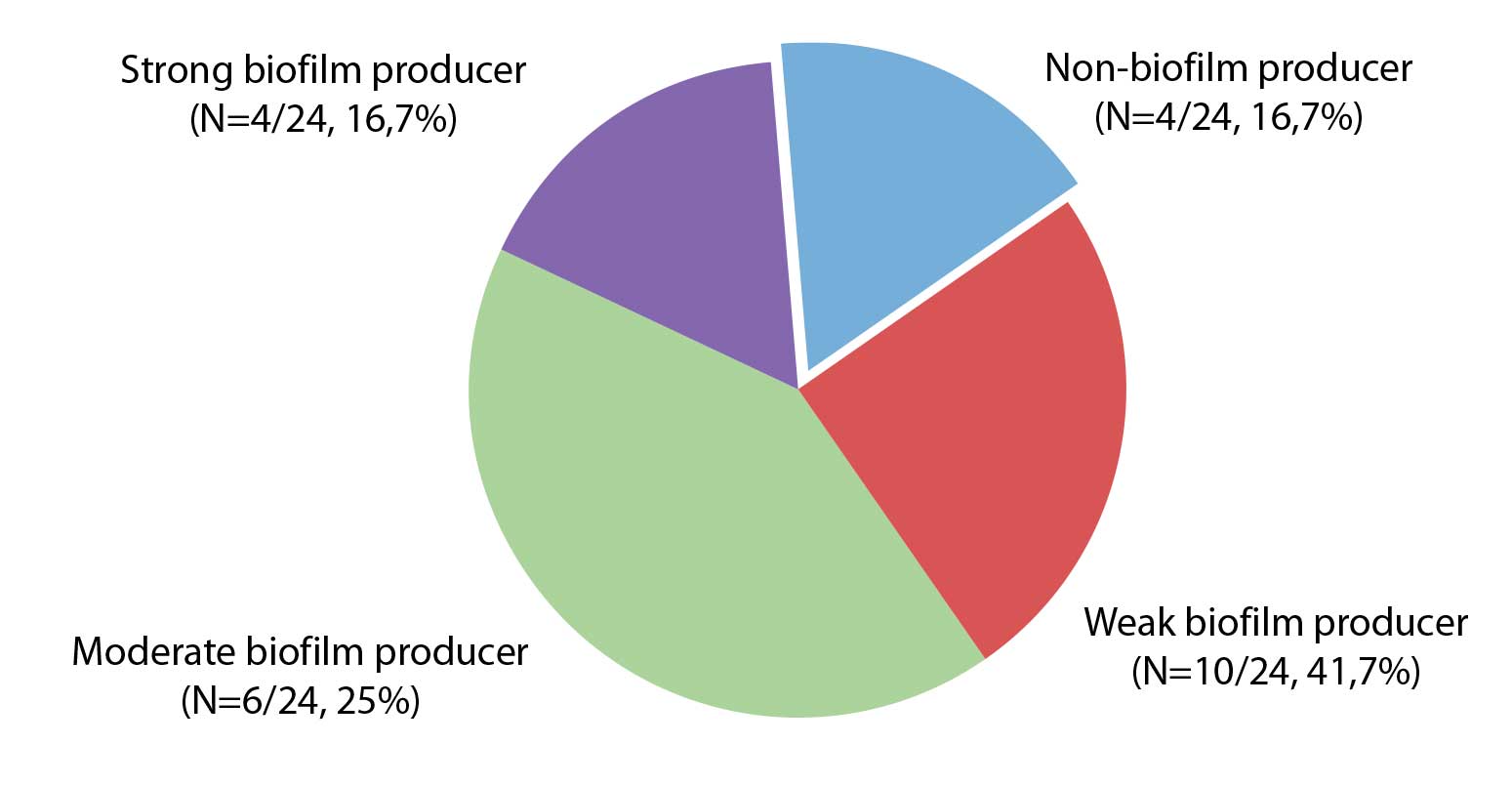
The results of testing the biofilm production ability of S. maltophilia isolates are shown in Figure 2. It was found that 83.3% (N=20/24) of the isolates are biofilm producers.
Figure 2. Biofilm producing capacity in the tested isolates of Stenotrophomonas maltophilia
Numerous studies have shown the importance of biofilm production in the development of S. maltophilia infections (Flores-Treviño et al., 2019; Pompilio et al., 2021). In addition to the stimulation of bacterial growth and biofilm formation, the DSF family of molecules induces the synthesis of L1 and L2 beta-lactamases, thus participating in the regulation of resistance mechanisms to beta-lactam antibiotics, in a still insufficiently elucidated manner (Alcaraz et al., 2019). In our study the frequency of strong biofilm producers was higher (N=4/24, 16.7%) compared to the research conducted in Serbia in the period 2013-2015 (N=7/88, 7.9%) (Madi et al., 2016)which has recently been recognized as a globally multi-drug resistant organism. The aim of this study was genotyping and physiological characterization of Stenotrophomonas maltophilia isolated in a large, tertiary care pediatric hospital in Belgrade, Serbia, hosting the national reference cystic fibrosis (CF. The same study notes a higher frequency of moderate biofilm producers (42%) compared to the results of this study (25%) (Madi et al., 2016)which has recently been recognized as a globally multi-drug resistant organism. The aim of this study was genotyping and physiological characterization of Stenotrophomonas maltophilia isolated in a large, tertiary care pediatric hospital in Belgrade, Serbia, hosting the national reference cystic fibrosis (CF. A study conducted in Iran in the period 2018-2019 observed only 28.2% of weak biofilm producers, while this phenotype dominates our research (50%) (Bostanghadiri et al., 2021)biofilm production, and the presence of biofilm genes among the S. maltophilia clinical isolates. A total of 85 clinical isolates of S. maltophilia were collected from patients referred to several hospitals. Susceptibility to antibiotics was investigated by disc diffusion method according to the guidelines of the Clinical and Laboratory Standards Institute (CLSI.
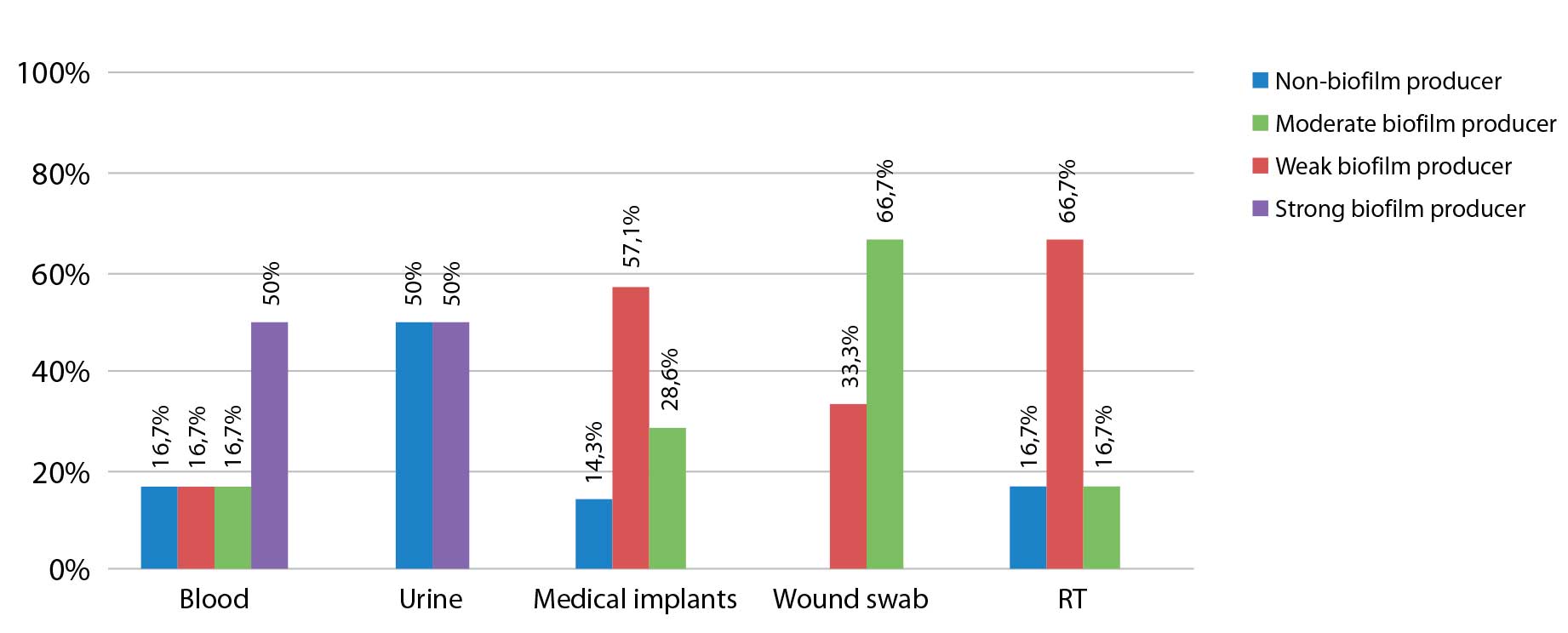
The description of the origin of the tested isolates in relation to their ability to produce biofilm is given in Figure 3. The most isolates (75%) that are strong biofilm producers were isolated from blood sample. Half of the strains (N=3/6, 50%) that moderately produce biofilm were isolated from the tip of the aspiration catheter. Our study detected 4 weak biofilm producers of respiratory tract origin, and this association is confirmed by a Mexican study (José Mauricio Del Río-Chacón et al., 2024). It was no statistically significant association observed between the ability to produce biofilm and the origin of isolates in this study (p>0.05).
Figure 3. Distribution of biofilm capacity groups of the tested Stenotrophomonas maltophilia isolates in samples.
RT – respiratory tract samples: tracheal swab, bronchoalveolar lavage (BAL), tracheal aspirate; medical implants: central venous catheter (CVC), aspiration catheter, chest drain
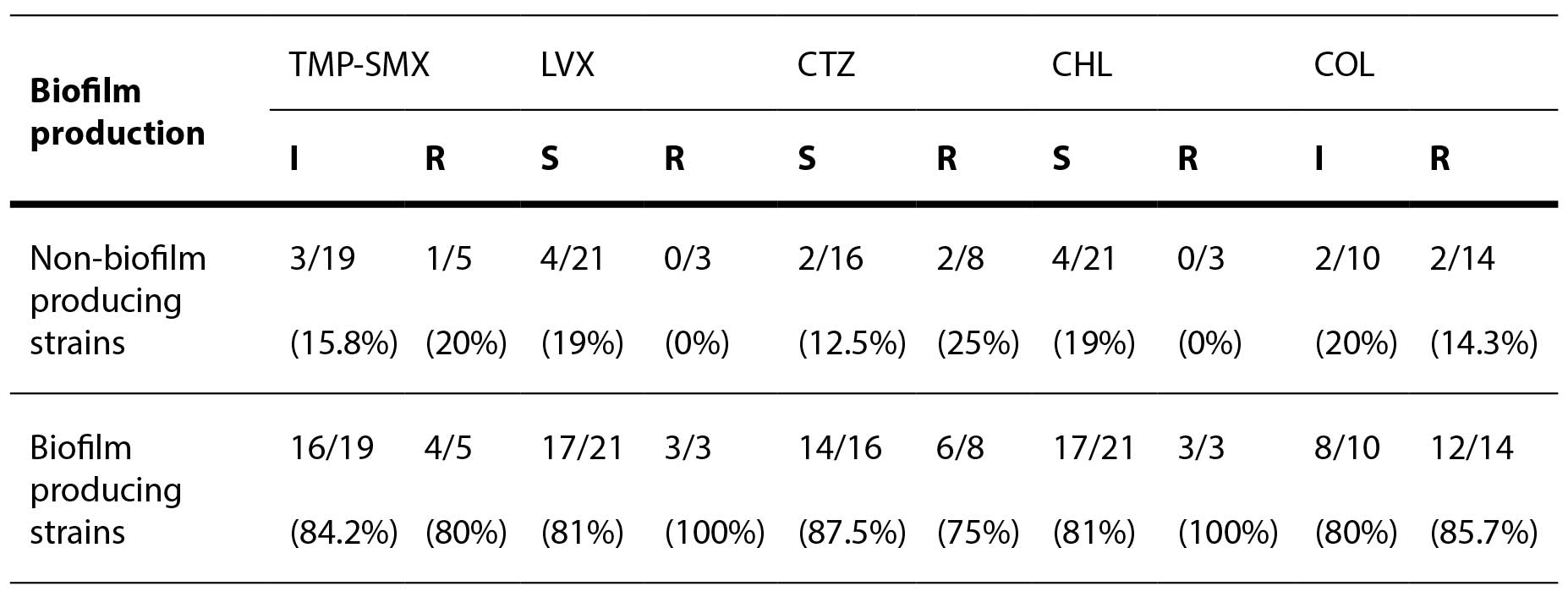
The distribution of sensitivity to antibiotics according to the biofilm production ability of the tested isolates is shown in Table 1.
Table 1. Distribution of antimicrobial susceptibility due to biofilm producing ability of the tested strains of Stenotrophomonas maltophilia
TMP-SMX – trimethoprim-sulfamethoxazole; LVX – levofloxacin; CTZ – ceftazidime; CHL – chloramphenicol; COL – colistin
Two out of three isolates (66.7%) in which colistin was the only therapeutic option were weak biofilm producers. An international study confirmed that strains sensitive to colistin, ceftazidime, and levofloxacin better produce biofilm. (Pompilio et al., 2021). No statistically significant association was observed between the ability to produce biofilm and resistance to certain antibiotics (p>0.05). Research from Thailand found that non-resistance isolates are significantly better biofilm producers compared to MDR strains (p< 0.05). This may be explained by the fact that S. maltophilia isolates from hospital environments relied on the expression of virulence factors for survival, rather than the ability to produce biofilms (Yinsai et al., 2023)information on the epidemiology and characteristics of this bacterium, especially in Thailand, is rarely found. This study aimed to determine the demographic, genotypic, and phenotypic characteristics of S. maltophilia isolates from Maharaj Nakorn Chiang Mai Hospital, Thailand. A total of 200 S. maltophilia isolates were collected from four types of clinical specimens from 2015 to 2016 and most of the isolates were from sputum. In terms of clinical characteristics, male and aged patients were more susceptible to an S. maltophilia infection. The majority of included patients had underlying diseases and were hospitalized with associated invasive procedures. The antimicrobial resistance profiles of S. maltophilia isolates showed the highest frequency of resistance to ceftazidime and the lower frequency of resistance to chloramphenicol, levofloxacin, trimethoprim/sulfamethoxazole (TMP/SMX.
The main limitation of this research is the small number of isolates collected over a long period of time (2017-2022). S. maltophilia isolates included in our study were sent randomly and voluntarily from regional laboratories. Also, additional genotyping of the isolates would provide additional information on the clonal relationship of the isolates, the basis of resistance to the tested antibiotics and the presence of genes responsible for biofilm formation.
4. Conclusion
The emergence of resistance to trimethoprim-sulfamethoxazole, observed in 20.8% of S. maltophilia isolates in our study, represents a significant problem. The presence of multidrug resistant strains in which colistin is the only therapeutic option in 12.5% of cases is an alarming data. The possibility of biofilm formation observed in 83.3% of isolates is of particular importance considering that it promotes the onset of infection and contributes to antimicrobial resistance. The previously presented data indicate that it is necessary to examine in detail the frequency and genetic basis of resistance to antibiotics in a larger number of clinical isolates of S. maltophilia in our country.
Acknowledgments: This research was supported by the Science Fund of the Republic of Serbia, Grant No. 7042, Tracking antimicrobial resistance in diverse ecological niches—one health perspective—TRACE
Conflicts of Interest: The authors declare no conflicts of interest
Literature
Abbott, I. J., Slavin, M. A., Turnidge, J. D., Thursky, K. A., & Worth, L. J. (2011). Stenotrophomonas maltophilia: Emerging disease patterns and challenges for treatment. Expert Review of Anti-Infective Therapy, 9(4), 471–488.
Alcaraz, E,. García, C,. Friedman, L,. Passerini de Rossi, B. (2019). The rpf/DSF signalling system of Stenotrophomonas maltophilia positively regulates biofilm formation, production of virulence-associated factors and β-lactamase induction. FEMS Microbiology Letters, 366, Fnz069, 4(1), 88–100.
Aoki, W., Uwamino, Y., Niida, N., Kubota, H., & Kamoshita, Y. (2025). Journal of Global Antimicrobial Resistance Cefiderocol susceptibility of Stenotrophomonas maltophilia species complex and carbapenem-resistant Pseudomonas aeruginosa isolates from blood cultures at a university hospital in Tokyo , Japan. Journal of Global Antimicrobial Resistance, 44, 251–255.
Biagi, M., Tan, X., Wu, T., Jurkovic, M., Vialichka, A., Meyer, K., Mendes, R. E., & Wenzler, E. (2020). Activity of potential alternative treatment agents for Stenotrophomonas maltophilia isolates nonsusceptible to levofloxacin and/or Trimethoprim-sulfamethoxazole. Journal of Clinical Microbiology, 58(2), 1–9.
Bostanghadiri, N., Ardebili, A., Ghalavand, Z., Teymouri, S., Mirzarazi, M., Goudarzi, M., Ghasemi, E., & Hashemi, A. (2021). Antibiotic resistance, biofilm formation, and biofilm-associated genes among Stenotrophomonas maltophilia clinical isolates. BMC Research Notes, 14(1), 4–9.
Brooke, J. S. (2012). Stenotrophomonas maltophilia: An emerging global opportunistic pathogen. Clinical Microbiology Reviews, 25(1), 2–41.
Cercenado, E., Cardenoso, L., Penin, R., Longshaw, C., Henriksen, A. S., & Pascual, A. (2021). In vitro activity of cefiderocol and comparators against isolates of Gram-negative bacterial pathogens from a range of infection sources: SIDERO‑WT‑2014−2018 studies in Spain. Journal of Global Antimicrobial Resistance, 26, 292–300.
Chang, Y. T., Lin, C. Y., Chen, Y. H., & Hsueh, P. R. (2015). Update on infections caused by Stenotrophomonas maltophilia with particular attention to resistance mechanisms and therapeutic options. Frontiers in Microbiology, 6(SEP), 1–20.
Chung, H. S., Kim, K., Hong, S. S., Hong, S. G., Lee, K., & Chong, Y. (2015). The sul1 gene in Stenotrophomonas maltophilia with high-level resistance to trimethoprim/sulfamethoxazole. Annals of Laboratory Medicine, 35(2), 246–249.
Cillóniz, C., Dominedò, C., & Torres, A. (2019). Multidrug Resistant Gram-Negative Bacteria in Community-Acquired Pneumonia. Critical Care, 23(1):79.
Çıkman, A., Parlak, M., Bayram, Y., Güdücüoğlu, H., & Berktaş, M. (2016). Antibiotics resistance of Stenotrophomonas maltophilia strains isolated from various clinical specimens. African Health Sciences, 16(1), 149–152.
Clinical and Laboratory Standards Institute (CLSI). (2025). Performance standards for antimicrobial susceptibility testing (35th ed., CLSI standard M100)
EUCAST: Breaking point tables for interpretation of MICs and zone diameters. (2025). European Committee on Antimicrobial Susceptibility Testing Breakpoint tables for interpretation of MICs and zone diameters European Committee on Antimicrobial Susceptibility Testing Breakpoint tables for interpretation of MICs and zone diameters.
Falagas, M. E., Kastoris, A. C., Vouloumanou, E. K., & Dimopoulos, G. (2009). Community-acquired Stenotrophomonas maltophilia infections: A systematic review. European Journal of Clinical Microbiology and Infectious Diseases, 28(7), 719–730.
Fan, X., Chen, F., Ruan, Z., & Li, L. (2025). Therapeutic challenges in managing Stenotrophomonas maltophilia bloodstream infection in a renal dysfunction patient: a case study and literature review. Frontiers in Cellular and Infection Microbiology, 15, 1–7.
Flores-Treviño, S., Bocanegra-Ibarias, P., Camacho-Ortiz, A., Morfín-Otero, R., Salazar-Sesatty, H. A., & Garza-González, E. (2019). Stenotrophomonas maltophilia biofilm: its role in infectious diseases. Expert Review of Anti-Infective Therapy, 17(11), 877–893.
Galani, I., Kontopidou, F., Souli, M., Rekatsina, P. D., Koratzanis, E., Deliolanis, J., & Giamarellou, H. (2008). Colistin susceptibility testing by Etest and disk diffusion methods. International Journal of Antimicrobial Agents, 31(5), 434–439.
García-León, G., Ruiz de Alegría Puig, C., García de la Fuente, C., Martínez-Martínez, L., Martínez, J. L., & Sánchez, M. B. (2015). High-level quinolone resistance is associated with the overexpression of smeVWX in Stenotrophomonas maltophilia clinical isolates. Clinical Microbiology and Infection, 21(5), 464–467.
Gibb, J., & Wong, D. W. (2021). Antimicrobial treatment strategies for Stenotrophomonas maltophilia: A focus on novel therapies. Antibiotics, 10(10), 1226.
Gil-Gil, T., Martínez, J. L., & Blanco, P. (2020). Mechanisms of antimicrobial resistance in Stenotrophomonas maltophilia: a review of current knowledge. Expert Review of Anti-Infective Therapy, 18(4), 335–347.
Gülmez, D., Çakar, A., Şener, B., Karakaya, J., & Hasçelik, G. (2010). Comparison of different antimicrobial susceptibility testing methods for Stenotrophomonas maltophilia and results of synergy testing. Journal of Infection and Chemotherapy, 16(5), 322–328.
Hafiz, T. A., Aldawood, E., Albloshi, A., Alghamdi, S. S., Mubaraki, M. A., Alyami, A. S., & Aldriwesh, M. G. (2022). Stenotrophomonas maltophilia Epidemiology, Resistance Characteristics, and Clinical Outcomes: Understanding of the Recent Three Years’ Trends. Microorganisms, 10(12).
Río-Chacón JMD, Rojas-Larios F, Bocanegra-Ibarias P, Salas-Treviño D, Espinoza-Gómez F, Camacho-Ortiz A, Flores-Treviño S. (2024). Biofilm Eradication of Stenotrophomonas maltophilia by Levofloxacin and Trimethoprim-Sulfamethoxazole. Japanese Journal of Infectious Diseases, 77(4), 213-219.
Juhász, E., Krizsán, G., Lengyel, G., Grósz, G., Pongrácz, J., & Kristóf, K. (2014). Infection and colonization by Stenotrophomonas maltophilia: Antimicrobial susceptibility and clinical background of strains isolated at a tertiary care centre in Hungary. Annals of Clinical Microbiology and Antimicrobials, 13(1), 1–7.
Juhász, E., Pongrácz, J., Iván, M., & Kristóf, K. (2015). Antibiotic susceptibility of sulfamethoxazole-trimethoprim resistant Stenotrophomonas maltophilia strains isolated at a tertiary care centre in Hungary. Acta Microbiologica et Immunologica Hungarica, 62(3), 295–305.
Katsube, T., Echols, R., & Wajima, T. (2019). Pharmacokinetic and Pharmacodynamic Profiles of Cefiderocol, a Novel Siderophore Cephalosporin. Clinical Infectious Diseases, 69(Suppl 7), S552–S558.
Lin, Q., Zou, H., Chen, X., Wu, M., Ma, D., Yu, H., Niu, S., & Huang, S. (2021). Avibactam potentiated the activity of both ceftazidime and aztreonam against S. maltophilia clinical isolates in vitro. BMC microbiology, 21(1), 60..
Madi, H., Lukić, J., Vasiljević, Z., Biočanin, M., Kojić, M., Jovčić, B., & Lozo, J. (2016). Genotypic and phenotypic characterization of Stenotrophomonas maltophilia strains from a pediatric tertiary care hospital in Serbia. PLoS ONE, 11(10), 1–13.
McCreary, E. K., Heil, E. L., & Tamma, P. D. (2021). New perspectives on antimicrobial agents: Cefiderocol. Antimicrobial Agents and Chemotherapy, 65(8), 1–14.
Mikhailovich, V., Heydarov, R., Zimenkov, D., & Chebotar, I. (2024). Stenotrophomonas maltophilia virulence: a current view. In Frontiers in Microbiology, 15, 1385631.
Milne, K. E. N., & Gould, I. M. (2012). Combination antimicrobial susceptibility testing of multidrug-resistant Stenotrophomonas maltophilia from cystic fibrosis patients. Antimicrobial Agents and Chemotherapy, 56(8), 4071–4077.
Mojica, M. F., Humphries, R., Lipuma, J. J., Mathers, A. J., Rao, G. G., Shelburne, S. A., Fouts, D. E., Van Duin, D., & Bonomo, R. A. (2022). Clinical challenges treating Stenotrophomonas maltophilia infections: An update. JAC-Antimicrobial Resistance, 4(3), 1–19.
Moskowitz, S. M., Garber, E., Chen, Y., Clock, S. A., Tabibi, S., Miller, A. K., Doctor, M., & Saiman, L. (2010). Colistin susceptibility testing: Evaluation of reliability for cystic fibrosis isolates of Pseudomonas aeruginosa and Stenotrophomonas maltophilia. Journal of Antimicrobial Chemotherapy, 65(7), 1416–1423.
Pompilio, A., Ranalli, M., Piccirilli, A., Perilli, M., Vukovic, D., Savic, B., Krutova, M., Drevinek, P., Jonas, D., Fiscarelli, E. V., Assanti, V. T. G., Tavío, M. M., Artiles, F., & Di Bonaventura, G. (2021). Biofilm formation among Stenotrophomonas maltophilia isolates has clinical relevance: The ANSELM prospective multicenter study. Microorganisms, 9(1), 1–25.
Rodríguez, C. H., Nastro, M., Calvo, J. L., Fariña, M. E., Dabos, L., & Famiglietti, A. (2014). In vitro activity of colistin against Stenotrophomonas maltophilia. Journal of Global Antimicrobial Resistance, 2(4), 316–317.
Sader, H. S., Castanheira, M., Arends, S. J. R., & Doyle, T. B. (2025). Antimicrobial susceptibility of Stenotrophomonas maltophilia from United States medical centers (2019–2023). Antimicrobial agents and chemotherapy, 69(4), e0012425.
Sader, H. S., Duncan, L. R., Arends, S. J. R., & Carvalhaes, C. G., and Castanheira, M. (2020). Antimicrobial Activity of Aztreonam-Avibactam and Comparator Agents When Tested against a Large Collection of Contemporary Stenotrophomonas maltophilia Isolates from Medical Centers Worldwide. Antimicrobial agents and chemotherapy, 64(11), e01433-20.
Samonis, G., Karageorgopoulos, D. E., Maraki, S., Levis, P., Dimopoulou, D., Spernovasilis, N. A., Kofteridis, D. P., & Falagas, M. E. (2012). Stenotrophomonas maltophilia infections in a general hospital: Patient characteristics, antimicrobial susceptibility, and treatment outcome. PLoS ONE, 7(5).
Sánchez, M. B., & Martínez, J. L. (2010). SmQnr contributes to intrinsic resistance to quinolones in Stenotrophomonas maltophilia. Antimicrobial Agents and Chemotherapy, 54(1), 580–581.
Sánchez, M. B., & Martínez, J. L. (2015). The efflux pump SmeDEF contributes to trimethoprim-sulfamethoxazole resistance in Stenotrophomonas maltophilia. Antimicrobial Agents and Chemotherapy, 59(7), 4347–4348.
Stepanović, S., Vuković, D., Hola, V., Di Bonaventura, G., Djukić, S., Ćirković, I., & Ruzicka, F. (2007). Quantification of biofilm in microtiter plates: Overview of testing conditions and practical recommendations for assessment of biofilm production by staphylococci. Journal of Pathology, Microbiology and Immunology – the APMIS journal, 115(8), 891–899.
Toleman, M. A., Bennett, P. M., Bennett, D. M. C., Jones, R. N., & Walsh, T. R. (2007). Global emergence of trimethoprim/sulfamethoxazole resistance in Stenotrophomonas maltophilia mediated by acquisition of sul genes. Emerging Infectious Diseases, 13(4), 559–565.
Gajdács, M., & Urbán, E. (2019). Epidemiological Trends and Resistance Associated with Stenotrophomonas maltophilia Bacteremia: A 10-Year Retrospective Cohort Study in a Tertiary-Care Hospital in Hungary. Diseases (Basel, Switzerland), 7(2), 41
Wang, Y., He, T., Shen, Z., & Wu, C. (2018). Antimicrobial Resistance in Stenotrophomonas spp. Microbiology Spectrum, 6(1).
Yero, D., Huedo, P., Conchillo-Solé, O., Martínez-Servat, S., Mamat, U., Coves, X., Llanas, F., Roca, I., Vila, J., Schaible, U. E., Daura, X., & Gibert, I. (2020). Genetic Variants of the DSF Quorum Sensing System in Stenotrophomonas maltophilia Influence Virulence and Resistance Phenotypes Among Genotypically Diverse Clinical Isolates. Frontiers in Microbiology, 11(June), 1–15.
Yinsai, O., Deeudom, M., & Duangsonk, K. (2023). Genotypic Diversity, Antibiotic Resistance, and Virulence Phenotypes of Stenotrophomonas maltophilia Clinical Isolates from a Thai University Hospital Setting. Antibiotics, 12(2).
Závora, J., Adámková, V., Studená, A., & Kroneislová, G. (2025). In Vitro Activity of Cefiderocol and Aztreonam/Avibactam Against Gram-Negative Non-Fermenting Bacteria: A New Strategy Against Highly Antibiotic-Resistant Infectious Agents. Antibiotics, 14(8), 1–13.