1. Introduction
Raspberries represent one of the most economically important berry crops and are among the principal export products of the Serbian fruit production and processing sector. Owing to the high quality of the fruit, favorable agroecological conditions, and strong demand in both domestic and international markets, raspberry production holds a prominent position in Serbian agriculture and food industry (Nedeljković et al. 2024; Kljajić et al. 2025). According to the Statistical Office of the Republic of Serbia, raspberry production in Serbia reached approximately 94,051 t in 2024, while the preliminary value for 2025 was 82,577 t. Compared with the latest clearly available FAOSTAT-based estimate of world raspberry production (940,979 t in 2023), these values correspond approximately to 10.00% and 8.78%, respectively. The biological characteristics of raspberry cultivation, its high economic profitability, favorable climatic conditions, and the increasing demand for fresh and frozen berries have all contributed to the continued growth of this sector (Kljajić et al. 2025). However, the expansion of raspberry production must be accompanied by continuous attention to food safety. Raspberries are commonly consumed fresh or frozen, often without any virus-inactivating treatment before consumption, which makes the control of microbiological hazards particularly important (Nasheri et al. 2019). In such products, food safety concerns are not limited to bacterial contamination, since foodborne viruses may remain undetected in the absence of visible spoilage while retaining infectivity during storage and distribution.
Among the viral hazards associated with soft fruits, human norovirus and hepatitis A virus (HAV) are of particular concern. Human noroviruses are recognized as the leading cause of acute gastroenteritis worldwide, affecting all age groups and producing especially severe consequences in vulnerable populations such as young children, the elderly, and immunocompromised individuals (Carlson et al. 2024). Within the human norovirus group, genogroups I and II (GI and GII) are particularly relevant in food virology, since they are responsible for the great majority of human infections and represent the principal analytical targets in foodborne outbreak investigations and surveillance systems (Carlson et al. 2024). In outbreak settings, GII strains are generally more frequently reported than GI strains (Verhoef et al. 2015). Hepatitis A virus remains another important foodborne pathogen and is generally considered more clinically severe than norovirus because of its hepatic involvement (Olaimat et al. 2024). Both viruses are transmitted primarily by the fecal–oral route and may enter the food chain through contaminated irrigation or wash water, contaminated harvesting equipment, infected food handlers, or inadequate hygiene during postharvest handling (Koopmans and Duizer, 2004).
Berry fruits, particularly frozen raspberries and strawberries, have repeatedly been implicated in foodborne outbreaks caused mainly by norovirus and hepatitis A virus, including multiple norovirus outbreaks in Denmark and Finland and large hepatitis A outbreaks in Europe linked to frozen berry products (Le Guyader et al. 2004; Falkenhorst et al. 2005; Sarvikivi et al. 2012; Severi et al. 2015; Scavia et al. 2017; Enkirch et al. 2018). Such epidemiological findings have reinforced the importance of harmonized molecular methods for the detection of foodborne viruses in berry matrices. In Serbian food testing laboratories, molecular methods for the detection of norovirus and hepatitis A virus are routinely applied and are generally harmonized with the international standard ISO 15216:2019, Microbiology of the food chain — Horizontal method for determination of hepatitis A virus and norovirus in food using real-time RT-PCR — Part 2: Method for qualitative detection. This standard specifically targets norovirus genogroups I and II together with HAV, making these viral agents highly relevant for the virological examination of raspberries. At the same time, currently available RNA virus extraction procedures may be time-consuming, require specialized laboratory infrastructure, or involve steps that can compromise viral recovery during sample preparation (Velebit et al. 2025). Therefore, there is a continuing need for rapid, efficient, and reliable analytical workflows suitable for routine virological monitoring.
Accordingly, the aim of this study was to detect norovirus genogroup I (NoV GI), norovirus genogroup II (NoV GII), and hepatitis A virus (HAV) in raspberry samples by real-time RT-PCR performed in accordance with ISO 15216. Raspberry samples were collected from ten different producers in the municipalities of Arilje and Požega (Serbia) between January and December 2023. The application of a standardized analytical workflow was intended to generate data relevant to risk assessment, routine food safety surveillance, and the improvement of preventive measures in raspberry production and handling.
2. Materials and methods
2.1. Sample collection, handling and preparation
Raspberry samples were collected from ten producers in the municipalities of Arilje and Požega (Serbia), a region known for its favorable climatic conditions and long tradition of raspberry cultivation. Both fresh and frozen raspberries, intended for the domestic market and export, were analyzed for the presence of norovirus (NoV) and HAV during 2023 (January–December). After harvesting, raspberries were pre-cooled at −5 °C for up to 12 hours, followed by individual quick freezing (IQF) in a tunnel system and storage at −20 °C for up to 30 days before sorting and packaging. Composite samples consisted of approximately 300 g of fruit (one commercial package), from which 25 g subsamples (8–10 fruits) were used for analysis. Samples were properly packaged, labelled, and transported to the Laboratory for Molecular Methods in Food Analysis. In the laboratory, raspberries were weighed without prior washing, homogenization, or cutting to preserve fruit integrity. Only samples of adequate quality were analyzed, while those showing visible mould or condensation were excluded. Each analytical sample (25 ± 0.3 g) was transferred into a sterile 400 mL filter bag for further processing.
2.2. Virus Concentration from Soft Fruit
Virus concentration was performed according to ISO 15216-2:2019, using the polyethylene glycol (PEG) precipitation method for soft fruit matrices.
Prior to extraction, 10 ± 0.5 μL of process control virus (MS2 bacteriophage) was added directly to the raspberry surface. Then, 40 ± 1 mL of TGBE buffer and ≥30 units of pectinase (from Aspergillus niger) were added. Samples were incubated at room temperature with continuous mixing (~60 oscillations/min) for 20 minutes.
During incubation, the pH of the eluate was monitored and, if necessary, adjusted to pH 9.5 ± 0.5 using NaOH. After incubation, the eluate was transferred into a 50 mL centrifuge tube and centrifuged at 10,000 × g for 30 minutes at 5 °C.
The supernatant was transferred to a new tube, and the pH was adjusted to 7.0 ± 0.5 using HCl. Virus precipitation was performed by adding 0.25 volumes of 5× PEG/NaCl solution, followed by incubation with mixing and centrifugation at 10,000 × g for 30 minutes at 5 °C.
The resulting pellet was resuspended in 500 μL phosphate-buffered saline (PBS). An additional purification step was performed by adding a chloroform/butanol mixture, followed by vortexing, incubation, and centrifugation. The aqueous phase containing viral RNA was then transferred to a new tube for further processing.
Extracts were either analysed immediately or stored at 5 °C (≤24 hours), −15 °C (≤6 months), or −70 °C for long-term storage.
2.3. Viral RNA Extraction
Viral RNA extraction was performed using the foodproof® Sample Preparation Kit IV (HYGIENA Diagnostic, Germany) according to the manufacturer’s instructions.
Viral particles were lysed by incubation with lysis buffer, releasing viral RNA. The addition of carrier RNA (tRNA) enhanced RNA binding to the silica membrane, thereby improving extraction efficiency and assay sensitivity.
After addition of absolute ethanol, RNA selectively bound to silica membrane fibers in filter tubes. Bound RNA was purified through two washing steps followed by centrifugation to remove potential PCR inhibitors. Finally, RNA was eluted using an elution buffer and collected for downstream analysis.
2.4. Detection of Viral RNA by Real-Time RT-PCR
Detection of norovirus genogroups I (GI) and II (GII) and hepatitis A virus (HAV) was performed using one-step real-time reverse transcription PCR (RT-PCR).
Amplification was carried out using the foodproof® Norovirus (GI, GII) plus Hepatitis A Virus Detection Kit® (HYGIENA Diagnostic, Germany). The assay contains specific primers and hydrolysis TaqMan probes targeting viral genomic regions, as well as a process control for result validation.
Primer and probe sequences used in the assay comply with the ISO 15216:2019 standard (Table 1-3).
Table 1. Primer sequences of HAV
Table 2. Primer sequences of GI
Table 3. Primer sequences of GII
2.5. Process Control
To prevent false-negative results caused by potential PCR inhibition from complex food matrices, a process control (MS2 bacteriophage) was added to each sample before virus concentration.
During reverse transcription, viral RNA was converted into complementary DNA (cDNA), which served as the template for amplification.
The multiplex assay enabled simultaneous detection of all three viral targets and the process control in a single reaction. The analytical sensitivity of the assay was 3–10 copies per reaction.
Amplification reactions were performed on an Applied Biosystems™ 7500 Real-Time PCR System (Thermo Fisher Scientific, USA).
Table 4. Primer sequences of GII
3. Results
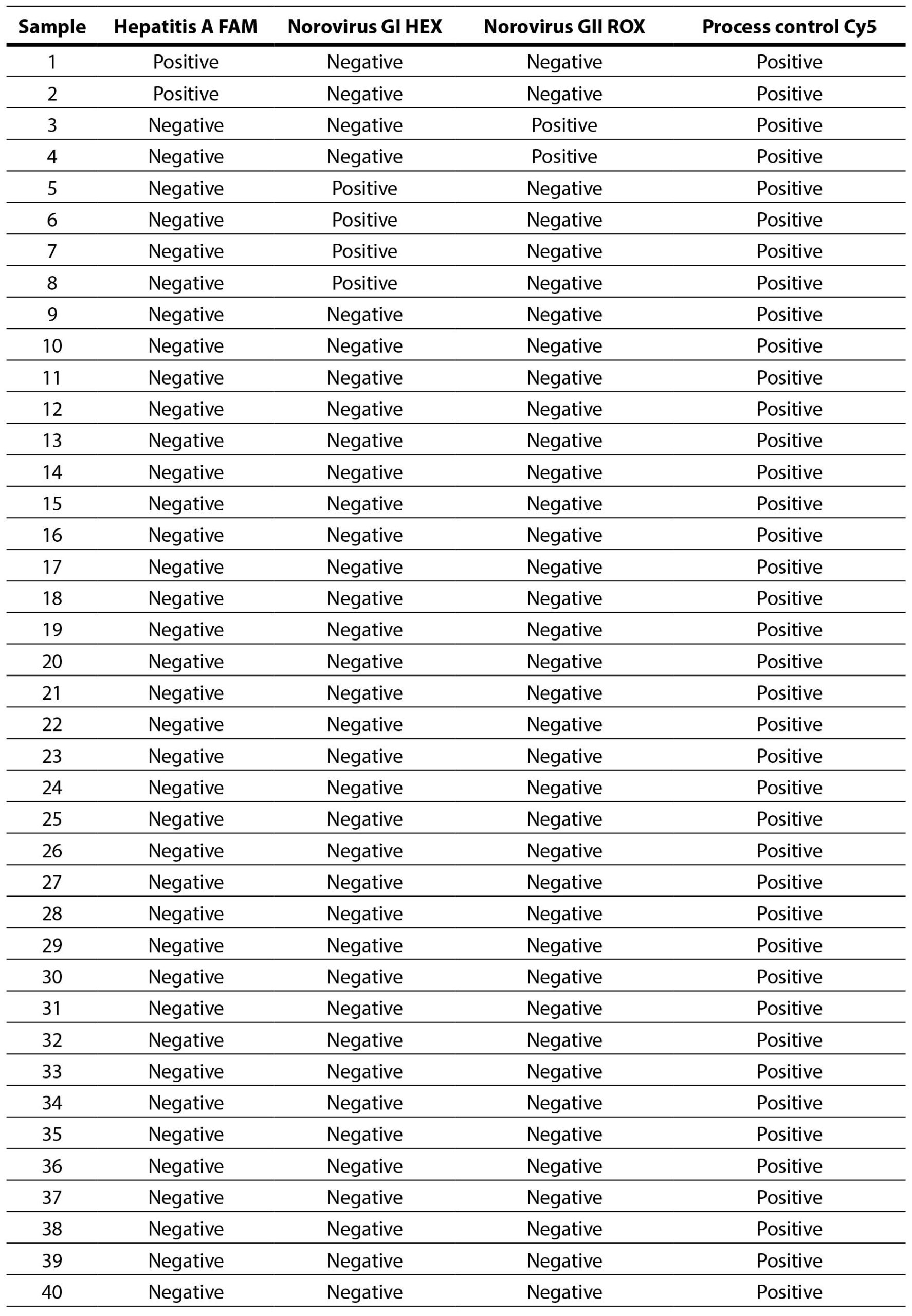
It was determined that, out of a total of 40 samples, 32 samples were negative for the presence of NoV GI, NoV GII, and HAV, 4 samples were positive for NoV GI (negative for NoV GII and HAV), 2 samples were positive for NoV GII (negative for NoV GI and HAV), and 2 samples were positive for HAV (negative for NoV GI and NoV GII).
The table below presents an overview of the RT-PCR reaction results for 40 samples, obtained using a multiplex kit for the detection of Norovirus group I (GI) and II (GII), hepatitis A virus (genotypes I, II, and III), as well as the process/internal control.
Table 5. Overview of RT-PCR reaction results
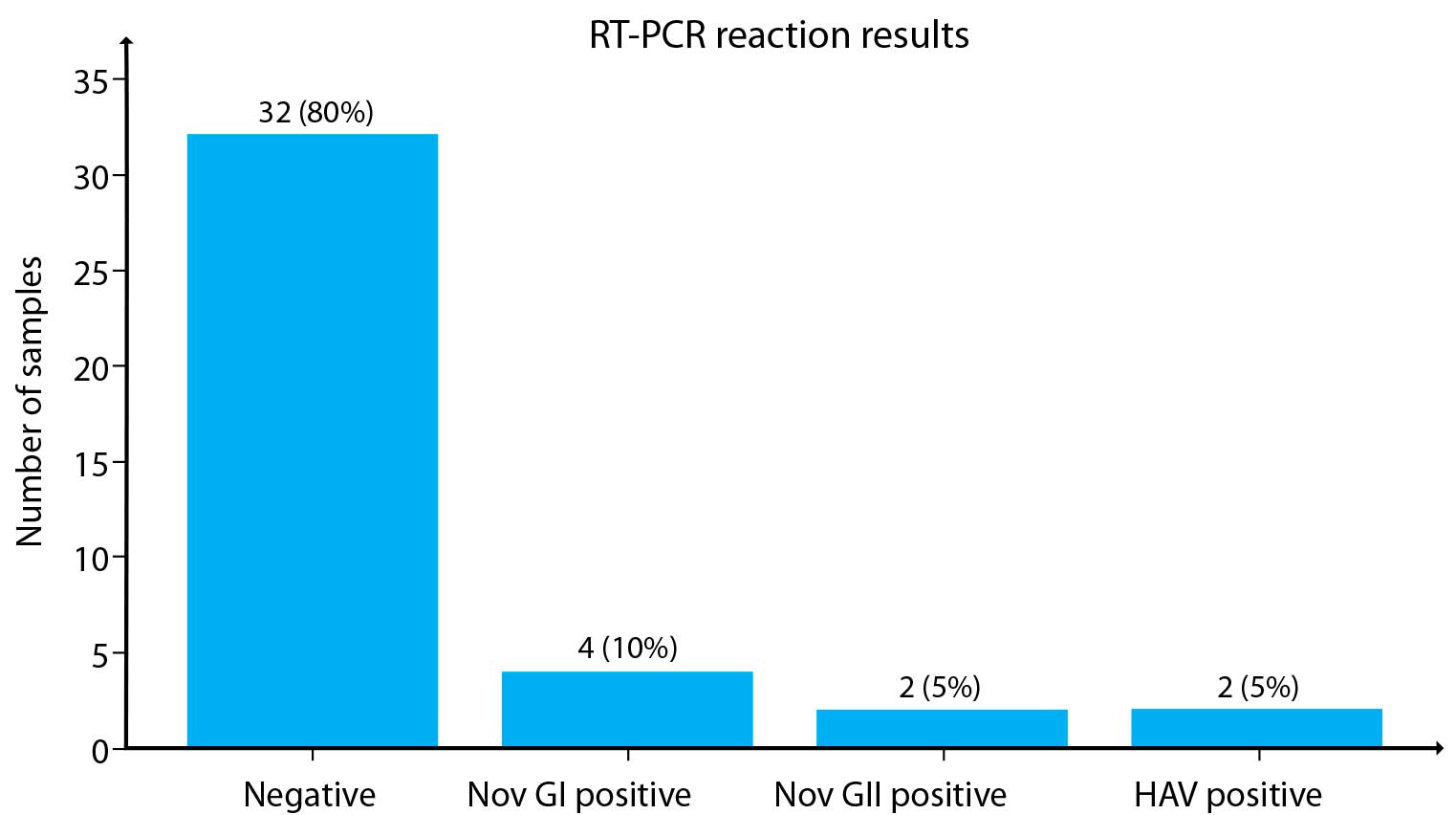
The graphical presentation provides an overview of the positive and negative RT-PCR reaction results.
Figure 1. Presentation of RT-PCR reaction results
Eighty percent of the samples were negative for all three targets, 5% were positive for HAV, 5% were positive for NoV GII, and 10% were positive for NoV GI (Figure 1).
4. Discusssion
Previous studies have clearly shown that NoV and HAV can persist in fruit matrices, including both fresh and frozen berries, which represents an important challenge for food safety surveillance and public health (Rodríguez-Lázaro et al. 2012). The persistence of enteric viruses in berries is influenced by several factors, including temperature, pH, matrix composition, and water activity, while freezing itself is generally regarded as a preservation process rather than an effective viral inactivation step (Bozkurt et al.,2021). Experimental and review data indicate that HAV can remain infectious on frozen berries for prolonged periods, and that freezing and frozen storage often result in only limited reductions in viral infectivity, thereby allowing contaminated products to remain a potential source of infection during distribution and consumption (Rzezutka & Cook, 2004).
The present results support the relevance of molecular surveillance of berry fruits for norovirus genogroups I and II and HAV. The detection of these viral targets in both fresh and frozen raspberry samples indicates that contamination may occur under different production, handling, storage, and distribution conditions, and confirms that both product categories require virological attention. These findings are consistent with earlier reports demonstrating the occurrence of enteric viruses in berry supply chains and highlighting multiple contamination routes, including contaminated irrigation water, infected food handlers, and postharvest handling environments (Maunula et al. 2013; Mäde et al. 2013; Bozkurt et al. 2021).
The use of real-time RT-PCR in this study proved appropriate for routine virological screening, particularly because molecular methods remain the basis of internationally approved detection protocols for norovirus and HAV in berries and other soft fruit. At the same time, the broader literature emphasizes that molecular detection, although highly sensitive and indispensable for surveillance, does not by itself resolve all analytical challenges associated with heterogeneous contamination, low viral loads, and the inability of RT-PCR alone to directly determine infectivity. For this reason, routine monitoring should be interpreted as one component of a broader preventive strategy that includes strict hygienic control during primary production, harvesting, processing, and distribution (Maunula et al. 2013).
An additional point of importance is that positive findings in raspberries are not only relevant from a public health perspective, but may also have substantial economic implications. Berry-associated outbreaks of NoV and HAV have repeatedly triggered regulatory concern, product recalls, and intensified control measures in international trade (Severi et al. 2015; Scavia et al. 2017; Tavoschi et al. 2015). Because Serbia is an important raspberry-producing country, the implementation of reliable and standardized virological monitoring may contribute both to consumer protection and to maintaining confidence in export chains.
Looking ahead, further improvement of analytical workflows for virus detection in berries remains necessary. Recent literature continues to emphasize the need for faster, more robust, and more informative detection strategies, including improved recovery methods, better characterization of low-level contamination, and the integration of advanced molecular tools for strain-level investigation where appropriate (Velebit et al. 2025). Such developments could strengthen outbreak tracing, improve source attribution, and support more effective risk management across the berry production chain.
- Conclusion
In conclusion, the results of this study confirm the presence of NoV GI, NoV GII, and HAV in raspberry samples, demonstrating that raspberries may represent a relevant vehicle for foodborne viral contamination. The detection of these viruses in samples collected from different producers and during different periods of the year underlines the importance of continuous virological surveillance of both fresh and frozen berries. The application of a standardized real-time RT-PCR approach in accordance with ISO-based principles provides a suitable basis for routine food safety monitoring. Nevertheless, sustained improvement of preventive measures, sampling strategies, and analytical workflows remains essential for reducing viral risk, protecting public health, and supporting the safety and marketability of raspberry products.
Acknowledgments: This work was financially supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia under Agreement No. 451-03-34/2026-03/200116 for 2026.
Conflict of interest: The authors declare no conflict of interest.
References
Bozkurt, H., Phan-Thien, K.-Y., van Ogtrop, F., Bell, T., & McConchie, R. (2021). Outbreaks, occurrence, and control of norovirus and hepatitis A virus contamination in berries: A review. Critical Reviews in Food Science and Nutrition, 61(1), 116–138.
Carlson, K. B., Dilley, A., O’Grady, T., Johnson, J. A., Lopman, B., & Viscidi, E. (2024). A narrative review of norovirus epidemiology, biology, and challenges to vaccine development. npj Vaccines, 9, Article 103.
Enkirch, T., Eriksson, R., Persson, S., Schmid, D., Aberle, S. W., Löf, E., Wittesjö, B., Holmgren, B., Johnzon, C., Gustafsson, E. X., Svensson, L. M., Labbé Sandelin, L., Richter, L., Lindblad, M., Brytting, M., Maritschnik, S., Tallo, T., Malm, T., Sundqvist, L., & Lundberg Ederth, J. (2018). Hepatitis A outbreak linked to imported frozen strawberries by sequencing, Sweden and Austria, June to September 2018. Eurosurveillance, 23(41), 1800528.
Falkenhorst, G., Krusell, L., Lisby, M., Madsen, S. B., Böttiger, B., & Mølbak, K. (2005). Imported frozen raspberries cause a series of norovirus outbreaks in Denmark, 2005. Eurosurveillance, 10(9), E050922.2.
International Organization for Standardization. (2019). Microbiology of the food chain—Horizontal method for determination of hepatitis A virus and norovirus using real-time RT-PCR—Part 2: Method for detection (ISO Standard No. 15216-2:2019). ISO.
Kljajic, N., Pomianek, I., & Vuković, P. (2025). A production and economic analysis of raspberry and its impact on the sustainable development of rural areas: A special focus on the situation in Serbia and Poland. Economics of Agriculture, 72(2), 519–540.
Koopmans, M., & Duizer, E. (2004). Foodborne viruses: An emerging problem. International Journal of Food Microbiology, 90(1), 23–41.
Le Guyader, F. S., Mittelholzer, C., Haugarreau, L., Hedlund, K.-O., Alsterlund, R., Pommepuy, M., & Svensson, L. (2004). Detection of noroviruses in raspberries associated with a gastroenteritis outbreak. International Journal of Food Microbiology, 97(2), 179–186.
Maunula, L., Kaupke, A., Vasickova, P., Söderberg, K., Kozyra, I., Lazic, S., van der Poel, W. H. M., Bouwknegt, M., Rutjes, S., Willems, K. A., Moloney, R., D’Agostino, M., de Roda Husman, A. M., von Bonsdorff, C.-H., Rzeżutka, A., Pavlik, I., Petrovic, T., & Cook, N. (2013). Tracing enteric viruses in the European berry fruit supply chain. International Journal of Food Microbiology, 167(2), 177–185.
Mäde, D., Trübner, K., Neubert, E., Höhne, M., & Johne, R. (2013). Detection and typing of norovirus from frozen strawberries involved in a large-scale gastroenteritis outbreak in Germany. Food and Environmental Virology, 5(3), 162–168.
Nasheri, N., Vester, A., & Petronella, N. (2019). Foodborne viral outbreaks associated with frozen produce. Epidemiology and Infection, 147, e291.
Nedeljković, M., Ćosić, M., & Marin, F.-E. (2024). Raspberry production trends in Serbia. Proceedings of the International Conference on Business Excellence, 18(1), 3235–3241.
Olaimat, A. N., Taybeh, A. O., Al-Nabulsi, A., Al-Holy, M., Hatmal, M. M., Alzyoud, J., Aolymat, I., Abughoush, M. H., Shahbaz, H., Alzyoud, A., Osaili, T., Ayyash, M., Coombs, K. M., & Holley, R. (2024). Common and potential emerging foodborne viruses: A comprehensive review. Life, 14(2), 190.
Rodríguez-Lázaro, D., Cook, N., Ruggeri, F. M., Sellwood, J., Nasser, A., Nascimento, M. S. J., D’Agostino, M., Santos, R., Saiz, J. C., Rzezutka, A., Bosch, A., Gironés, R., Carducci, A., Muscillo, M., Kovač, K., Diez-Valcarce, M., Vantarakis, A., von Bonsdorff, C.-H., de Roda Husman, A. M., Hernández, M., & van der Poel, W. H. M. (2012). Virus hazards from food, water and other contaminated environments. FEMS Microbiology Reviews, 36(4), 786–814.
Rzezutka, A., & Cook, N. (2004). Survival of human enteric viruses in the environment and food. FEMS Microbiology Reviews, 28(4), 441–453.
Sarvikivi, E., Roivainen, M., Maunula, L., Niskanen, T., Korhonen, T., Lappalainen, M., & Kuusi, M. (2012). Multiple norovirus outbreaks linked to imported frozen raspberries. Epidemiology and Infection, 140(2), 260–267.
Scavia, G., Alfonsi, V., Taffon, S., Escher, M., Bruni, R., De Medici, D., Di Pasquale, S., Guizzardi, S., Cappelletti, B., Iannazzo, S., Losio, N. M., Pavoni, E., Decastelli, L., Ciccaglione, A. R., Equestre, M., Tosti, M. E., Rizzo, C., & National Italian Task Force on Hepatitis A. (2017). A large prolonged outbreak of hepatitis A associated with consumption of frozen berries, Italy, 2013–14. Journal of Medical Microbiology, 66(3), 342–349.
Severi, E., Verhoef, L., Thornton, L., Guzman-Herrador, B. R., Faber, M., Sundqvist, L., Rimhanen-Finne, R., Roque-Afonso, A. M., Ngui, S. L., Allerberger, F., Baumann-Popczyk, A., Muller, L., Parmakova, K., Alfonsi, V., Tavoschi, L., Vennema, H., Fitzgerald, M., Myrmel, M., Gertler, M., Ederth, J., Kontio, M., Vanbockstael, C., Mandal, S., Sadkowska-Todys, M., Tosti, M. E., Schimmer, B., O’Gorman, J., Stene-Johansen, K., Wenzel, J. J., Jones, G., Balogun, K., Ciccaglione, A. R., O’Connor, L., Vold, L., Takkinen, J., & Rizzo, C. (2015). Large and prolonged food-borne multistate hepatitis A outbreak in Europe associated with consumption of frozen berries, 2013 to 2014. Eurosurveillance, 20(29), 21192.
Tavoschi, L., Severi, E., Niskanen, T., Boelaert, F., Rizzi, V., Liebana, E., Gomes Dias, J., Nichols, G., Takkinen, J., & Coulombier, D. (2015). Food-borne diseases associated with frozen berries consumption: A historical perspective, European Union, 1983 to 2013. Eurosurveillance, 20(29), 21193.
Velebit, B., Janković, V., Velebit, M., Bošković, T., Jovanović, M., Wang, D., & Mišić, D. (2025). Detection of norovirus from berries in Serbia by digital PCR and NGS. Foods, 14(18), 3257.
Verhoef, L., Hewitt, J., Barclay, L., Ahmed, S. M., Lake, R., Hall, A. J., Lopman, B., Kroneman, A., Vennema, H., Vinjé, J., & Koopmans, M. (2015). Norovirus genotype profiles associated with foodborne transmission, 1999–2012. Emerging Infectious Diseases, 21(4), 592–599.