1. Introduction
Gram-positive bacteria, belonging to the genus Enterococcus, have a distinctive cell wall structure that allows them to survive adverse environmental conditions and gives them intrinsic resistance to a large number of antibiotics, including cephalosporins, aminoglycosides, macrolides, etc. (Khalil et al. 2025.). These bacteria are part of the gut microbiota and, hence, they serve as indicators of fecal contamination of drinking water. Due to the robustness of their cell wall, they can also survive in saline settings and are therefore commonly used for the detection of fecal contamination in marine waters (Peter et al. 2025).
Enterococci are bacteria of great importance within the One Health concept, as they are capable of infecting both humans and animals and can also survive on abiotic surfaces such as water and soil. Monitoring their antimicrobial resistance is extremely important, as they are members of the “ESKAPE” (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa and Enterobacter spp.) group of multidrug-resistant bacteria, which have a major impact on human and/or animal infections, especially as a frequent cause of intrahospital infections (Miller & Arias, 2024). Antibiotic resistance can spread through vertical and horizontal transfer of genetic material between bacteria of the same genus or genetically related bacteria; therefore, potentially multidrug-resistant strains can be easily transmitted from hospital environments into the external environment.
In addition to antibiotic resistance, the ability of Enterococcus spp. to produce biofilm plays a major role in their persistence in the external environment (Shiopu et al. 2023). Within these sessile communities, bacteria are protected from environmental stressors as well as from antibacterial substances. Biofilms can be established on human tissues, leading to chronic infections, or on abiotic surfaces in nutrient-poor environments, enabling long-term survival and persistence. Despite the clinical and environmental significance of biofilm formation, its assessment is not yet routinely performed in microbiological laboratories, even though its detection could be highly valuable in predicting the development of antimicrobial resistance during prolonged therapy for chronic infections.
This research aimed to determine the frequency of Enterococcus spp. in drinking water samples from poultry farms, as well as to analyse their antibiotic susceptibility, biofilm-forming ability, and the antibiofilm activity of commonly used biocides (bleach and hydrogen peroxide) at various concentrations.
2. Material and methods
2.1. Bacterial isolates
This study was conducted as part of the project “Public Health Risk of the Frequency and Antimicrobial Resistance of Pathogenic Bacteria in Drinking Water Originating from Poultry Farms” (No. 19.032/961-101/23), conducted during the period from 1 April 2024 to 1 May 2025. A total of 120 drinking water samples were collected from poultry farmsDetection of Enterococcus spp. was performed in accordance with the standard BAS EN ISO 7899-2, which involves filtration of water samples, cultivation on selective media, and identification based on characteristic colony morphology and biochemical tests. Isolates were stored under appropriate conditions for subsequent antimicrobial susceptibility and biofilm analyses.
2.2. Antimicrobial susceptibility testing
Antibiotic susceptibility testing of all Enterococcus isolates was performed in accordance with European Committee on Antimicrobial Susceptibility Testing (EUCAST) guidelines (EUCAST, 2024) using the disk diffusion method. Briefly, an inoculum of 0.5 McFarland units was inoculated onto Mueller–Hinton agar plates, after which the following antibiotic disks were applied to the dried plates: ampicillin, imipenem, nitrofurantoin, norfloxacin, and vancomycin. Plates were incubated at 35–37°C for 18–24 hours. Following overnight incubation, the diameters of inhibition zones were measured, and antibiotic susceptibility was interpreted as susceptible (S), susceptible, increased exposure (I), or resistant (R).
Susceptibility to ampicillin was used as an indicator for the susceptibility to amoxicillin-clavulanic acid, ampicillin-sulbactam, piperacillin, and imipenem. Additionally, norfloxacin was used as a screening agent for susceptibility to all fluoroquinolones except ofloxacin, which is not tested for Enterococcus spp.
Quality control was performed using reference strains to ensure accuracy and reproducibility of the results.
2.3. Biofilm production and antibiofilm activity of biocides testing
Biofilm formation by Enterococcus spp. was assessed using the microtiter plate method, as described by Stepanović et al., with slight modifications (2007). Briefly, bacterial isolates were first cultured overnight at 37 °C in Trypticase Soy Broth (TSB; Bio-Rad, United Kingdom) and subsequently diluted in fresh TSB medium to achieve a final concentration of 10⁶ CFU/mL. A volume of 100 µL of each bacterial suspension was dispensed into each well of a sterile 96-well microtiter plates and incubated at 37 °C for 24 h to allow biofilm formation. After incubation, the wells were gently emptied and washed three times with sterile phosphate-buffered saline (PBS) to remove non-adherent (planktonic) cells. The adherent cells were fixed and stained with 100 µL of 2% (w/v) crystal violet for 15 minutes. Excess stain was removed by washing the wells three times with PBS, followed by air drying at room temperature overnight. The retained crystal violet, representing the biofilm biomass, was then eluted using 100 µL of 33% (v/v) glacial acetic acid per well. Biofilm biomass was quantified by measuring the optical density (OD) at 570 nm with an automated microplate reader (ICN Flow Titertek Multiskan Plus Reader, Meckenheim, Germany). Wells containing only TSB were used as negative controls. The cut-off optical density (ODc) was calculated as the mean OD of the negative controls plus three standard deviations (SD). Based on OD measurements, isolates were categorized as follows:
non-biofilm producers (OD ≤ ODc),
weak biofilm producers (ODc < OD ≤ 2×ODc),
moderate biofilm producers (2×ODc < OD ≤ 4×ODc),
strong biofilm producers (OD > 4×ODc).
Following the initial 24-hour biofilm formation, the established Enterococcus biofilms were exposed to various concentrations of commonly used biocides for 5 minutes :
sodium hypochlorite (NaOCl, 0.004% and 0.4%)
hydrogen peroxide (H₂O₂, 0.0005% and 3%).
After biocide treatment, the wells were gently washed with sterile PBS to remove residual biocide and non-adherent cells. The biofilm biomass remaining after exposure was quantified spectrophotometrically using the same crystal violet staining procedure described in section 2.3. Percent reduction in biofilm biomass was calculated relative to untreated control wells to evaluate the antibiofilm efficacy of each biocide and concentration. This approach allows direct comparison of the effectiveness of different disinfectants against biofilm-associated Enterococcus spp. under controlled laboratory conditions.
2.4. Statistical analysis
Statistical analyses were performed using SPSS software version 20.0 (SPSS Inc., Chicago, IL, USA). Comparisons of the frequency of occurrence of categorical variables, including biofilm formation categories and biocide effectiveness, were conducted using Fisher’s exact test. Differences were considered statistically significant at p < 0.05.
Data from the biofilm and antibiofilm assays were presented as mean ± SD of at least three independent experiments.
3. Results
3.1. Bacterial strains
Out of the 120 drinking water samples examined from poultry farms, Enterococcus spp. (fecal enterococci) were detected in 16 samples (13.3%). Water contaminated in this manner is considered unsafe for human consumption as well as for use in broiler farms.
3.2. Antimicrobial susceptibility testing
Using the disk diffusion method, all 16 isolates were found to be susceptible to all five antibiotics tested.
3.3. Biofilm formation and antibiofilm activity of biocides
Of the 16 bacterial isolates tested, 10 (62.5%) were classified as strong biofilm producers, while the remaining 6 (37.5%) were considered moderate producers. None of tested isolates was classified as a non-producer.
The frequency of moderate and weak biofilm producers, as well as non-producers, following treatment with biocides is presented in Tables 1 and 2, respectively.
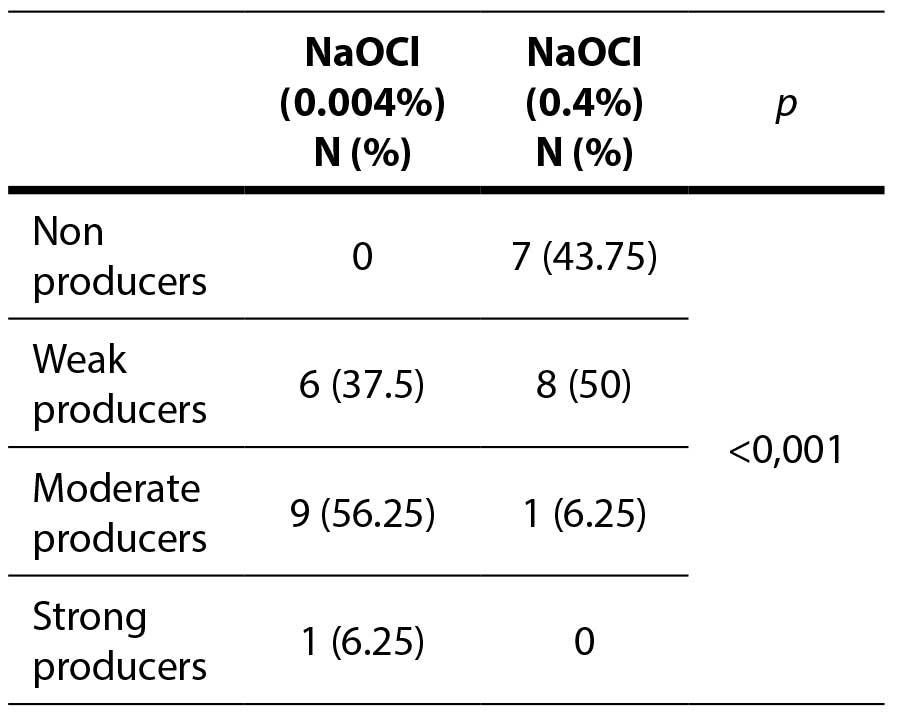
Table 1. Antibiofilm effect of the bleach against enterococcal biofilm
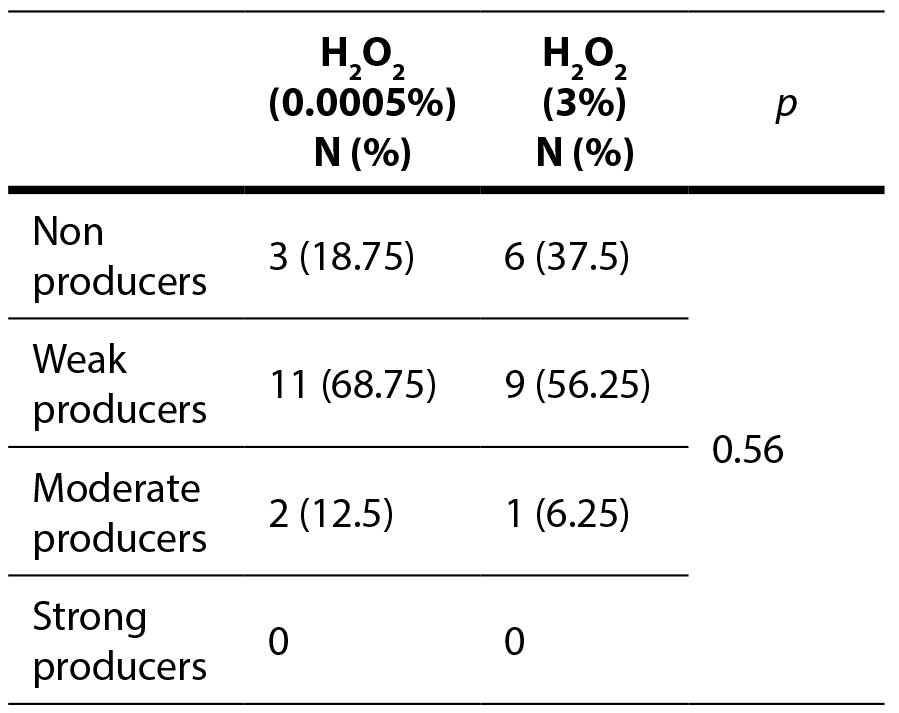
Table 2. Antibiofilm effect of the hydrogen peroxide against enterococcal biofilm
A statistically significant difference was observed between higher and lower concentrations of bleach, as well as when comparing the lower concentrations of biocides (p=0.007). No statistically significant difference was detected between higher and lower concentrations of hydrogen peroxide, nor when analyzing the effects of the higher concentrations of both biocides (p=1).
4. Discussion
Enterococci are highly significant bacteria with a broad range of hosts and the ability to persist in the environment, largely due to their strong biofilm-forming capacity. In our study, the isolates were susceptible to all antibiotics tested; however, they also exhibited a remarkable ability to form biofilms, which enables their prolonged survival in water pipes.
Microbiologically safe drinking water is a fundamental requirement for maintaining broiler health, and consequently human health as the end consumers. In our study, 13% of the samples were deemed unsatisfactory due to the presence of Enterococcus spp. This prevalence is lower than that observed in our previous study (Golic et al. 2023) and nearly half of that reported by other researchers in Austria and Canada. (Soares et al. 2023; Furtula et al. 2013). Our findings indicate that continuous collaboration between farmers, veterinarians, and microbiologists, combined with appropriate legal regulations and enforcement, can improve hygiene standards, benefiting both animal and human health. Additionally, it is important to note that our isolates were susceptible to all tested antibiotics, which is a very promising result, suggesting that multidrug-resistant strains have not been introduced into the drinking water of poultry farms. Similar results were noticed by Aboelseoud et al. (2021) in Egypt, who also showed the sensitivity of their enterococcal strains to the same antibiotics. In the future, antibiotic susceptibility of Enterococcus isolates from broiler gut samples and farm environmental samples should be investigated to assess the prevalence and resistance rates of these strains throughout the entire farm environment, as has been done in other studies. (Ribeiro et al. 2023; Furtula et al. 2013).
Biofilm production is one of the most important virulence factors that enables bacterial survival in water systems and the environment. Our isolates demonstrated a strong ability to form biofilms, consistent with findings from other studies. A limitation of this study was that bacterial detection was performed at the genus level rather than the species level. Grund et al. demonstrated that E. faecium and E. faecalis are more robust biofilm producers compared to E. cecorum (Grund et al. 2022). Furthermore, Wozniak et al. (2019) and Aboelseoud et al. (2021) confirmed our findings, showing that all strains possessed the ability to form biofilms.
Safe drinking water is routinely treated with disinfectants and biocides. The most commonly used agents are bleach (NaOCl) and hydrogen peroxide due to their effectiveness, affordability, and relatively eco-friendly properties. In our study, we demonstrated that NaOCl exhibited stronger antibiofilm activity at lower concentrations, while at higher concentrations, both disinfectants showed similar efficacy. It is important to note that neither biocide was able to eliminate established biofilms, a finding that is also supported by Castro et al. (2018). Additional measures should be implemented to prevent biofilm formation, as previously established biofilms cannot be fully eliminated, particularly in closed water supply systems and in hard-to-reach areas within water pipes.
In conclusion, although we observed a low prevalence of Enterococcus in drinking water and susceptibility of all isolates to the tested antibiotics, additional measures and analysis of samples from animals and the entire farm environment are needed to prevent the establishment of biofilms and sessile bacterial communities, which are extremely difficult to eradicate.
Acknowledgments: This research was funded by the Ministry of Scientific and Technological Development and Higher Education of the Republic of Srpska under the following grant number: 19.032/961-101/23.
Conflicts of Interest: The authors declare no conflicts of interest.
5. References
Aboelseoud, H., Ismael, E., Moustafa, G. Z., & Badawy, E. M. (2021). Hygienic studies on biofilms in drinking water systems in poultry farms: isolation, molecular identification, and antibiotic sensitivity. J Anim Health Prod 9(4), 443-454. http://dx.doi.org/10.17582/journal.jahp/2021/9.4.443.454
EUCAST. Breakpoint tables for interpretation of MICs and zone diameters Version 14.0. 2024. https://www.eucast.org/news-detail/breakpoint-table-140-2024-available-for-consultation-5-19-december-2023/
Furtula, V., Jackson, C. R., Farrell, E. G., Barrett, J. B., Hiott, L. M., & Chambers, P. A. (2013). Antimicrobial resistance in Enterococcus spp. isolated from environmental samples in an area of intensive poultry production. Int J Environ Res Pub Health, 10(3), 1020-103. https://doi.org/10.3390/ijerph10031020
Golić B., Knežević D., Pećana B. (2023): Assessment of the microbiological status of drinking water on animal farms. In XIV International Scientific Agricultural Symposium „Agrosym 2023“, Proceedings, 1086-1093.
Grund, A., Rautenschlein, S., & Jung, A. (2022). Detection of Enterococcus cecorum in the drinking system of broiler chickens and examination of its potential to form biofilms. Eur Poul Sci, 86, 1-15. https://doi.org/10.1399/eps.2022.346
Khalil, M. A., Alorabi, J. A., Al-Otaibi, L. M., Ali, S. S., & Elsilk, S. E. (2023). Antibiotic Resistance and Biofilm Formation in Enterococcus spp. Isolated from Urinary Tract Infections. Pathogens, 12(1), 34. https://doi.org/10.3390/pathogens12010034
Miller, W. R., & Arias, C. A. (2024). ESKAPE pathogens: antimicrobial resistance, epidemiology, clinical impact and therapeutics. Nat Rev Microbiol, 22(10), 598–616. https://doi.org/10.1038/s41579-024-01054-w.
Peter, P. O., Bertone, E., & Stewart, R. A. (2025). A systematic literature review of forecasting and predictive models for enterococci intrusion in aquatic ecosystems. Clean Water, 100063. https://doi.org/10.1016/j.clwat.2025.100063
Ribeiro, J., Silva, V., Monteiro, A., Vieira-Pinto, M., Igrejas, G., Reis, F. S., Barros, L., & Poeta, P. (2023). Antibiotic Resistance among Gastrointestinal Bacteria in Broilers: A Review Focused on Enterococcus spp. and Escherichia coli. Animals, 13(8), 1362. https://doi.org/10.3390/ani13081362
Santos Rosado Castro, M., da Silva Fernandes, M., Kabuki, D. Y., & Kuaye, A. Y. (2018). Biofilm formation of Enterococcus faecium on stainless steel surfaces: Modeling and control by disinfection agents. J Food Proc Engine, 41(3), e12663. https://doi.org/10.1111/jfpe.12663
Șchiopu, P., Toc, D. A., Colosi, I. A., Costache, C., Ruospo, G., Berar, G., Gălbău, Ș. G., Ghilea, A. C., Botan, A., Pană, A. G., Neculicioiu, V. S., & Todea, D. A. (2023). An Overview of the Factors Involved in Biofilm Production by the Enterococcus Genus. Int J Mol Sci, 24(14), 11577. https://doi.org/10.3390/ijms241411577
Soares, A. S., Miranda, C., Coelho, A. C., & Trindade, H. (2023). Occurrence of coliforms and enterococcus species in drinking water samples obtained from selected dairy cattle farms in Portugal. Agriculture, 13(4), 885. https://doi.org/10.3390/agriculture13040885;
Stepanović, S., Vuković, D., Hola, V., Di Bonaventura, G., Djukić, S., Cirković, I., & Ruzicka, F. (2007). Quantification of biofilm in microtiter plates: overview of testing conditions and practical recommendations for assessment of biofilm production by staphylococci. APMIS, 115(8), 891–899. https://doi.org/10.1111/j.1600-0463.2007.apm_630.x
Woźniak-Biel, A., Bugla-Płoskońska, G., Burdzy, J., Korzekwa, K., Ploch, S., & Wieliczko, A. (2019). Antimicrobial Resistance and Biofilm Formation in Enterococcus spp. Isolated from Humans and Turkeys in Poland. Microbial Drug Resistance, 25(2), 277–286. https://doi.org/10.1089/mdr.2018.0221.